The United States has witnessed a significant paradigm shift in the nicotine delivery and industrial interface markets. As a premier Mouth-to-Lung (MTL) Devices Supplier & Exporter, we recognize that the US market is no longer just about "cloud chasing." Instead, there is a sophisticated demand for high-resistance, low-wattage devices that mimic the traditional smoking experience—a core characteristic of MTL technology.
In states like California, Texas, and Florida, the vaping community has transitioned toward Pod Systems and Nicotine Salt compatible hardware. This shift is driven by the need for portability, discretion, and efficiency. Our export data shows that US consumers prioritize build quality and leak-proof designs, leading us to implement rigorous ISO-standard testing for every unit destined for North American shores.
Market Share Growth for MTL Pod Systems in the US (2023-2024)
FDA PMTA Compliance Readiness for Exported Hardware
Technical Support for our US B2B Partners
Shenzhen Droned Vape Co., Ltd. is a professional manufacturer specializing in the research, development, and production of a wide range of vaping devices. Our product portfolio includes Pod Systems, Vape Cartridges, Disposable Vapes, and various Vape Accessories. We are committed to delivering innovative and high-quality vaping solutions for global customers.

With years of industry experience and strong technical capabilities, Droned Vape focuses on product innovation, strict quality control, and customer-oriented services. Our products are widely exported to the United States, Canada, Russia, Thailand, South Africa, Kenya, Bahrain, the Middle East, and many other international markets, gaining trust and recognition from distributors and brand partners worldwide.
Our factory operates under standardized management systems and maintains a modern dust-free production workshop that meets international manufacturing standards. The company currently employs over 540 staff members, including:
In the US tech hubs, there is an increasing demand for MTL devices integrated with AI chips for temperature control and puff monitoring. Our latest MTL4516 series interfaces provide the precision required for these high-tech applications, ensuring safety and consistency.
Beyond recreational use, MTL (Mouth-to-Lung) principles are applied in medical inhalation devices. Our Endotracheal Tube Balloon Manufacturing Devices and Lung Assessment Mouthpieces are critical for US healthcare facilities, meeting the stringent quality requirements of the American medical sector.
Whether it's for the bustling streets of New York City or the industrial zones of the Midwest, our MTL devices are designed to withstand diverse environmental conditions while providing a premium user experience.
To ensure stable product quality and efficient production, our factory is equipped with advanced manufacturing equipment, including:

 Mtl4516 Switch Interface - US Industrial Standard
Mtl4516 Switch Interface - US Industrial Standard
 Mtl5511 Proximity Detector Interface - Export Grade
Mtl5511 Proximity Detector Interface - Export Grade
 Effective Lung Mucus Clearing Device - USA Healthcare
Effective Lung Mucus Clearing Device - USA Healthcare
 Medical Balloon Bonding Devices for US Manufacturers
Medical Balloon Bonding Devices for US Manufacturers
 Precision Inhalation Aerosol Release Device
Precision Inhalation Aerosol Release Device
 Disposable Mouthpieces for US Lung Assessment
Disposable Mouthpieces for US Lung Assessment
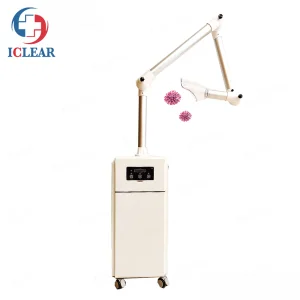 Mobile Dental Aerosol Suction - US Clinic Grade
Mobile Dental Aerosol Suction - US Clinic Grade
 Disposable Mouth to Mouth Face Shield - Safety First
Disposable Mouth to Mouth Face Shield - Safety First
 Air Source Treatment Units FRL for US Workshops
Air Source Treatment Units FRL for US Workshops
 Original Mtl Isolation Safety Barrier Mtl4511c
Original Mtl Isolation Safety Barrier Mtl4511c
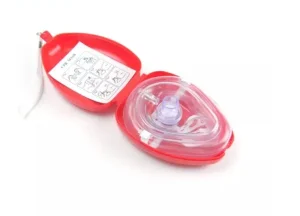 High Quality CPR Training Device - US Certification Ready
High Quality CPR Training Device - US Certification Ready
 Mtl 500mm Magnetic Fuel Level Sensor - US Logistics
Mtl 500mm Magnetic Fuel Level Sensor - US Logistics